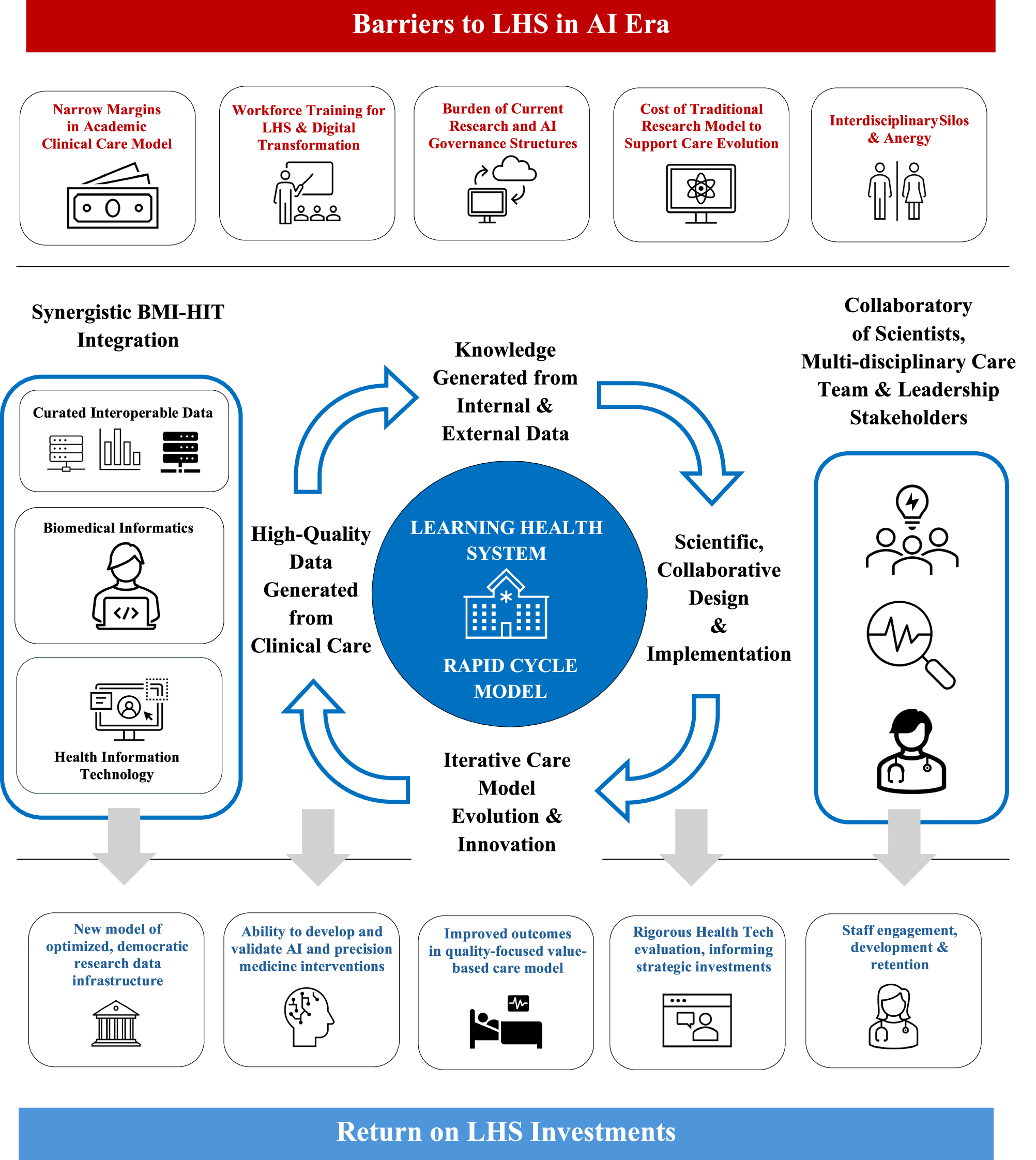
Pharmaceutical companies are increasingly recognizing the need to adapt their partnership strategies with multi-location health systems to enhance the efficiency of clinical trials. As the landscape of clinical research evolves, the integration of monitoring, supply chain management, and contractual agreements becomes crucial for successful collaborations with clinical research site networks. This shift is driven by the growing complexity of clinical trials, which require a more streamlined approach to site management and patient recruitment.
By embracing innovative partnership models, pharma companies can leverage the extensive resources and patient populations of multi-location health systems, thereby accelerating the trial process and improving data quality. This evolution not only addresses current regulatory and operational challenges but also positions companies to respond more effectively to future demands in the clinical research arena. As such, the ability to forge robust partnerships with health systems will be a key determinant of success in the competitive pharmaceutical landscape.
Use the database as your supply chain compass →