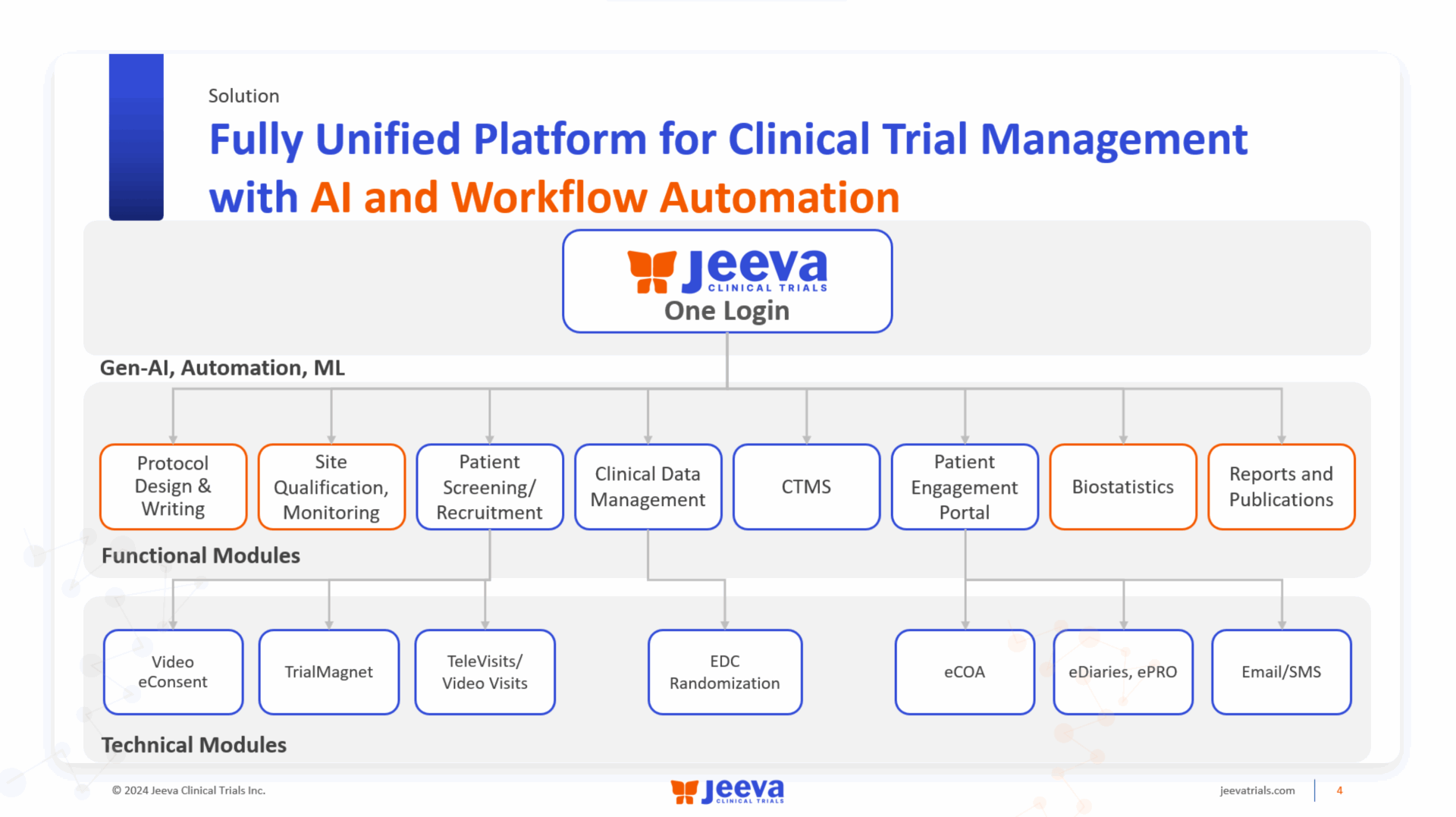
Clinical data management is undergoing a significant transformation as artificial intelligence (AI) automates electronic data capture (EDC) build processes, thereby reducing timelines and enhancing the overall quality of data management. This shift is not merely a technological upgrade; it represents a fundamental change in how data teams operate, allowing them to allocate more resources toward quality assurance and critical analysis rather than manual data entry and management tasks.
The integration of AI in data management processes is particularly timely, as the pharmaceutical industry faces increasing pressure to accelerate drug development while maintaining compliance with stringent regulatory standards. By streamlining data collection and management, AI tools can facilitate quicker decision-making and improve the accuracy of clinical trials, ultimately leading to faster market entry for new therapies.
As AI continues to evolve, its implications for regulatory, quality assurance, and supply chain professionals are profound. Embracing these technologies will not only enhance operational efficiency but also position organizations to better navigate the complexities of modern drug development and regulatory landscapes.
Get started today with Solo access →