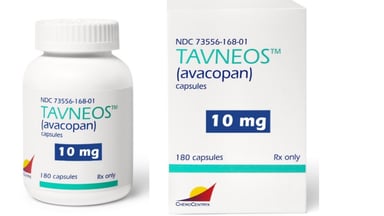
The U.S. Food and Drug Administration (FDA) has identified 76 cases of drug-induced liver injury associated with Amgen’s Tavneos, a treatment primarily indicated for the management of certain autoimmune conditions. Among these cases, eight fatalities were reported, raising significant concerns regarding the safety profile of the drug. This alarming data comes as the FDA continues to evaluate the risk-benefit balance of Tavneos in clinical settings.
The context of these findings highlights a growing scrutiny over the pharmacovigilance practices in the industry, particularly for drugs that have entered the market under accelerated approval pathways. The implications for Amgen could be substantial, potentially affecting not only the drug’s market position but also the company’s reputation in the pharmaceutical landscape. Regulatory bodies may impose stricter monitoring requirements, and healthcare providers will need to exercise increased caution when prescribing Tavneos, ensuring that patients are adequately informed of the associated risks.
Get started today with Solo access →