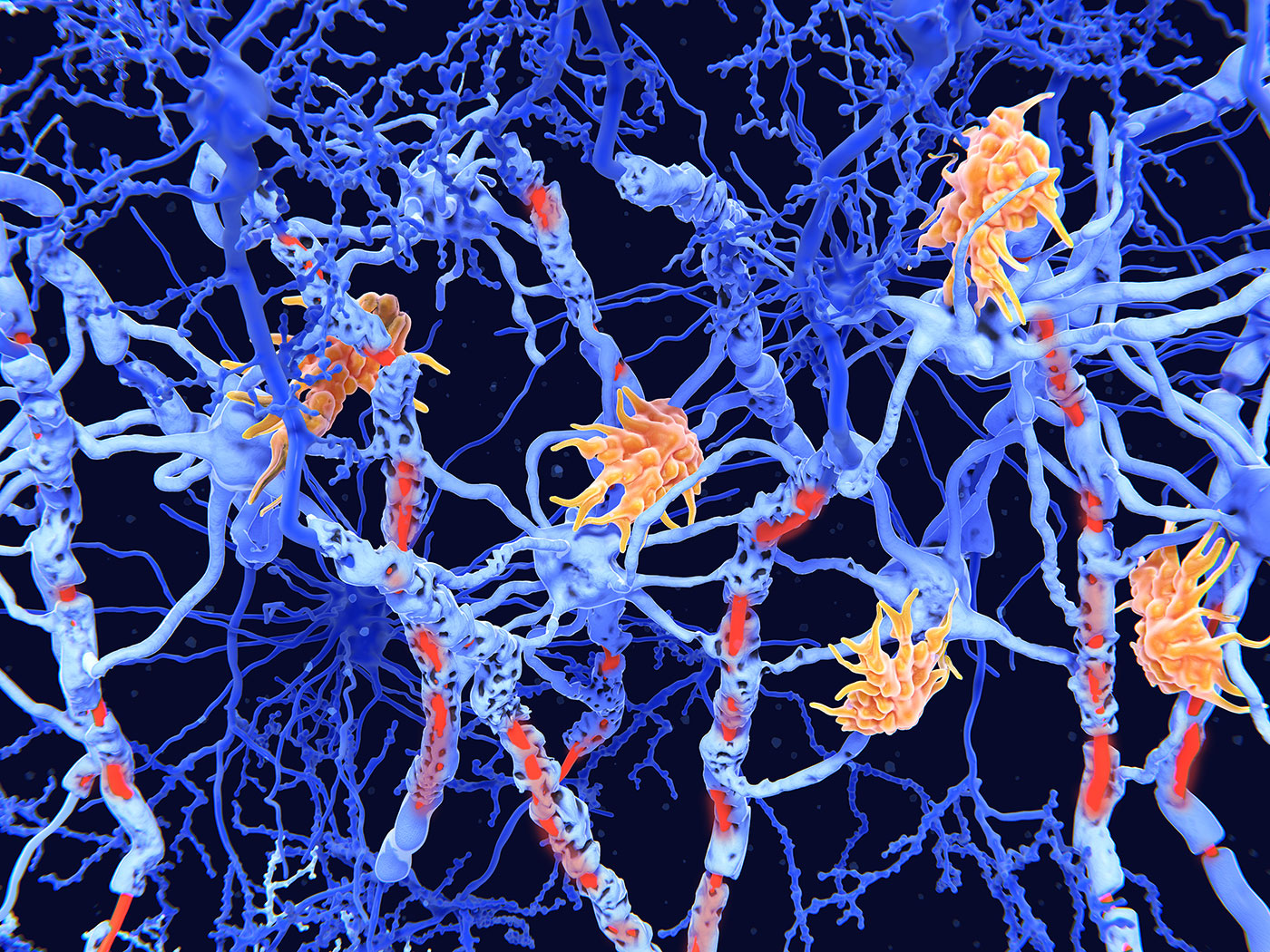
Researchers at University College London (UCL) have developed a hydrogel-based axon model that significantly enhances the study of myelin formation and the efficacy of remyelination therapies for multiple sclerosis (MS). This innovative model addresses a critical gap in drug development, as many candidates that show promise in traditional rigid lab settings fail in human trials due to the lack of physiological relevance. The new model accurately mimics the geometry and softness of native axons, which is crucial for oligodendrocyte function and myelin repair.
The UCL team utilized a microfabrication technique to create vertical micropillars from polyacrylamide hydrogel, allowing for precise control over the material’s properties. This advancement enables researchers to investigate drug candidates in a more realistic environment, potentially leading to better predictive outcomes in clinical settings. Senior author Emad Moeendarbary emphasized that the rigid models commonly used in laboratories may mislead drug discovery efforts, underscoring the importance of this new approach.
By faithfully replicating the physical characteristics of the human brain, this hydrogel-based model not only offers a more reliable platform for early drug testing but also facilitates high-content imaging and transcriptomic profiling. The implications of this work are significant, as it could streamline the development of effective remyelination therapies, ultimately improving treatment options for patients suffering from MS and related neurodegenerative conditions.
Open the full market picture for your next decision →