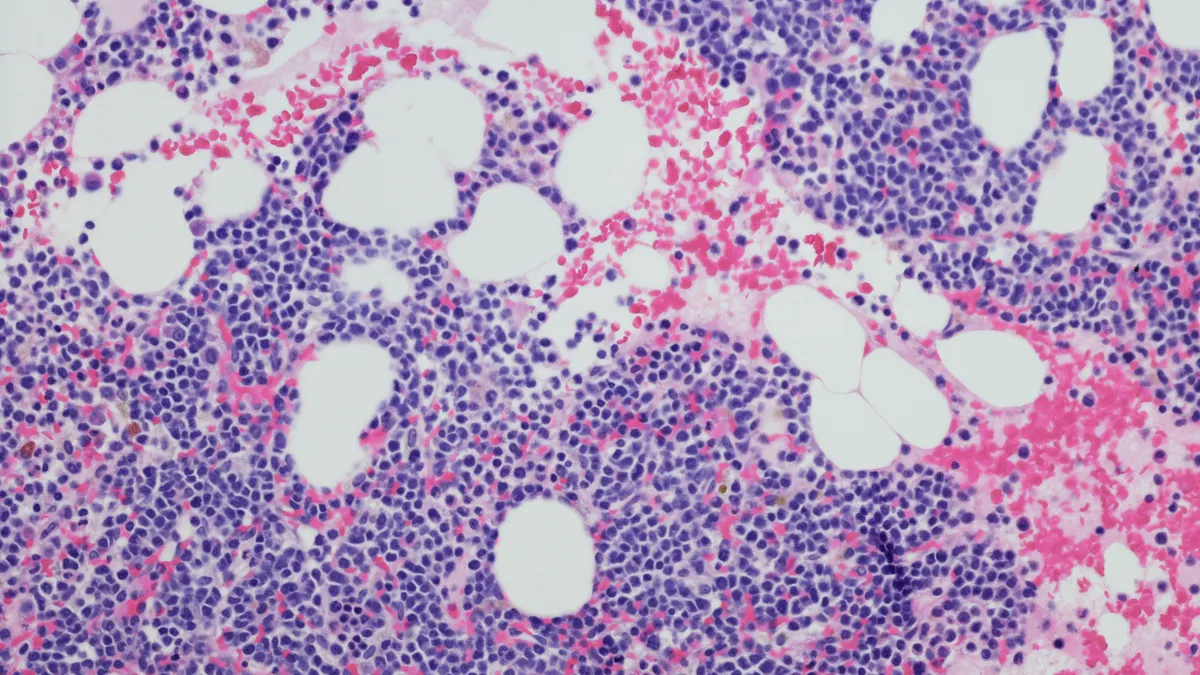
Johnson & Johnson has announced promising results from a study evaluating the earlier use of Tecvayli, a dual-targeting antibody, in patients with multiple myeloma. This development is significant as it positions Tecvayli for potential use in earlier treatment lines, rather than the later-line settings where it has primarily been utilized thus far.
The context of this advancement is critical; multiple myeloma remains a challenging blood cancer with limited treatment options in earlier stages. The ability to introduce Tecvayli sooner could enhance patient outcomes and alter the treatment landscape, providing a new avenue for healthcare providers in managing this complex disease.
The implications of these findings are substantial for the pharmaceutical industry, particularly for companies focused on hematologic malignancies. If further studies confirm these results, it could lead to expanded indications for Tecvayli, influencing market dynamics and potentially reshaping treatment protocols for multiple myeloma.
Use the database as your supply chain compass →