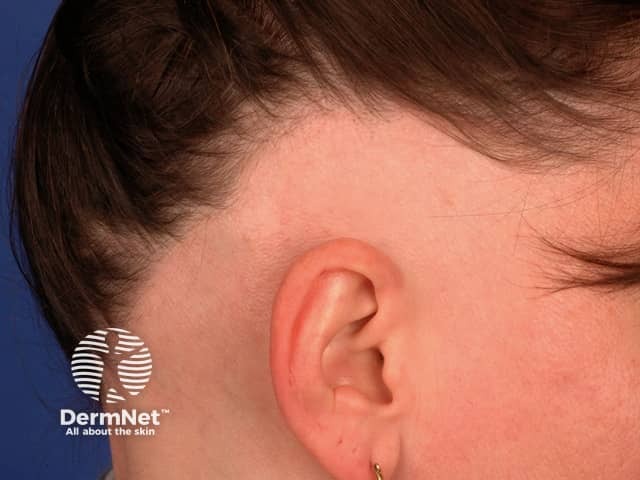
Eli Lilly announced plans to request a label expansion from the FDA for its immunology drug Olumiant, specifically targeting adolescents suffering from alopecia. This decision follows the release of promising long-term data that indicates the drug’s efficacy in this demographic. At the one-year follow-up, Lilly reported that 54% of participants experienced significant hair regrowth, underscoring the potential of Olumiant as a viable treatment option for younger patients facing this condition.
The move comes at a time when there is growing interest in effective therapies for alopecia, a condition that can have profound psychological and social implications for adolescents. By seeking to expand the indications for Olumiant, Lilly not only aims to address an unmet medical need but also positions itself competitively in the dermatological space, where treatment options remain limited. If approved, this could lead to increased market share and bolster Lilly’s portfolio in immunological treatments.
Get started today with Solo access →