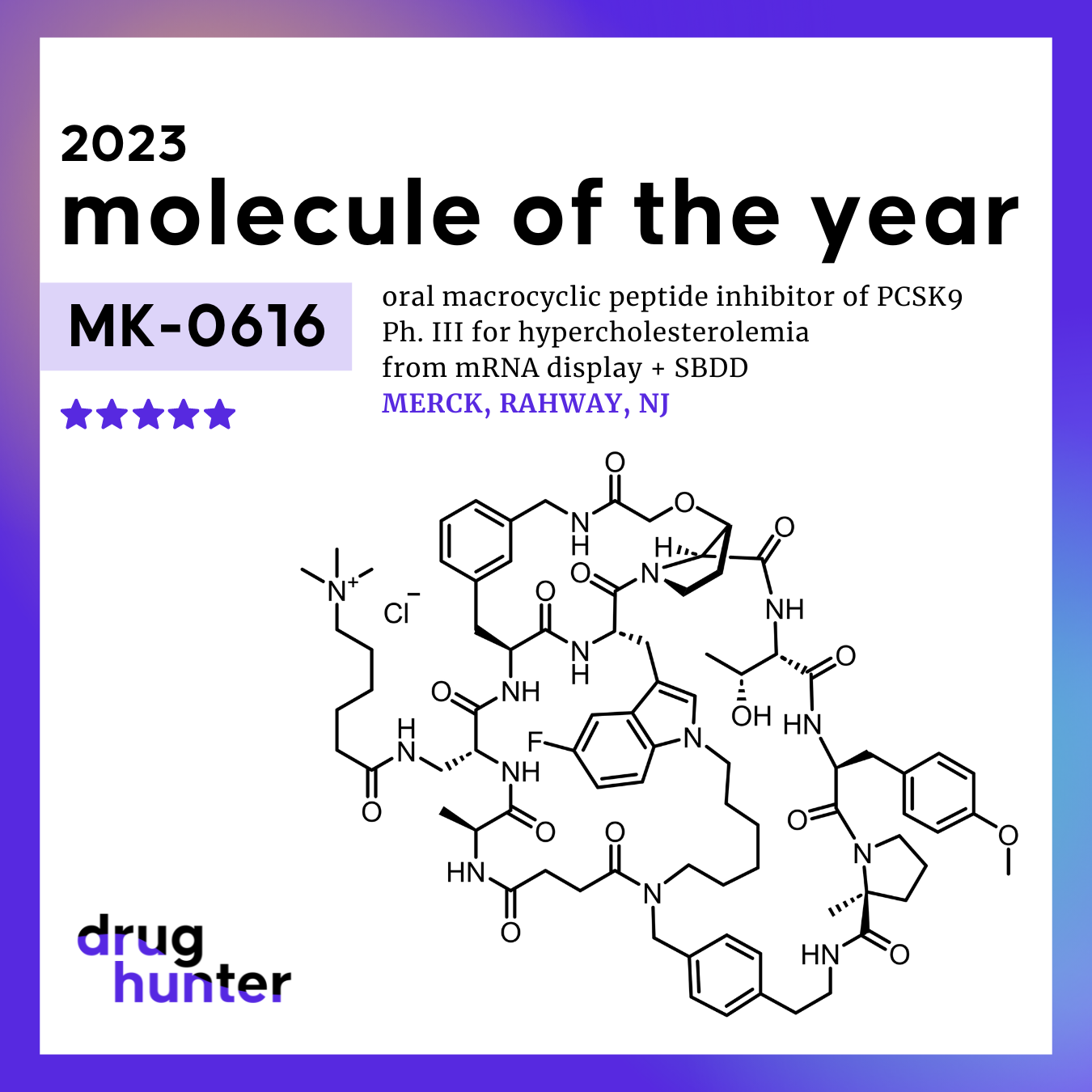
Merck has announced promising results from its latest clinical trials for an oral medication targeting PCSK9, a protein that plays a crucial role in cholesterol regulation. This development is significant as it addresses a longstanding challenge in making effective heart disease treatments more accessible to patients, particularly those who have struggled with injectable alternatives.
The FDA’s evolving regulatory landscape, under the leadership of Rick Pazdur, is also noteworthy. Pazdur’s new role could influence how quickly innovative therapies, such as Merck’s oral PCSK9 inhibitor, receive approval, potentially expediting access for patients in need. As the agency adapts to the increasing complexity of drug development, the implications for pharmaceutical companies are profound, necessitating a more agile approach to compliance and market entry.
With the recent focus on lowering drug prices, including GLP-1 medications, the interplay between regulatory decisions and market dynamics will be critical. Merck’s success could set a precedent for future heart disease therapies, reshaping the treatment landscape and impacting patient care significantly.
Open the full market picture for your next decision →