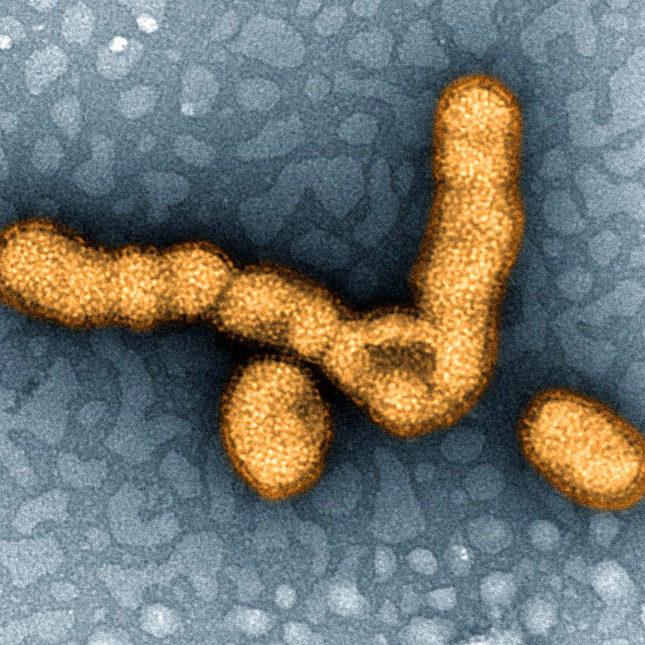
A recent case of swine flu reported in Spain has raised concerns among public health officials and pharmaceutical professionals alike. The case, which involves a patient exhibiting symptoms consistent with the H1N1 virus, highlights the ongoing risk of influenza outbreaks and the necessity for vigilant monitoring and rapid response strategies within the healthcare system.
This incident comes at a time when the pharmaceutical industry is under pressure to ensure adequate vaccine supply and distribution, particularly as seasonal flu approaches. The emergence of swine flu cases can complicate vaccine strategies, necessitating a reevaluation of current stockpiles and production capabilities. Regulatory bodies will need to assess the implications for vaccine efficacy and public health preparedness.
As the situation develops, it is crucial for B2B professionals in the pharma sector—particularly those in regulatory, QA/QC, and CMC roles—to stay informed and proactive. The potential for increased demand for vaccines and antiviral treatments may impact sourcing strategies and portfolio management, underscoring the need for agile responses to emerging health threats.
Open the full market picture for your next decision →