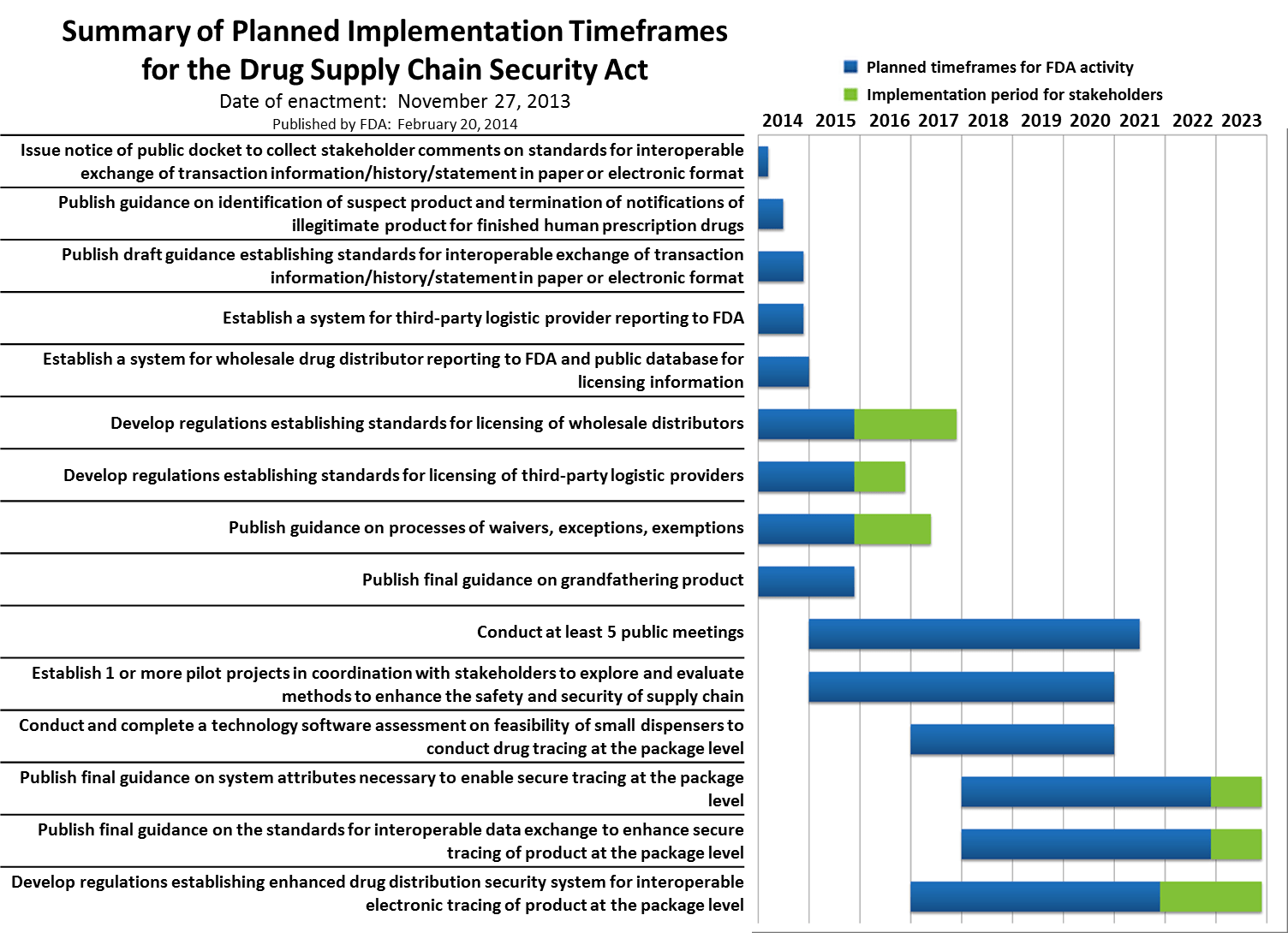
AstraZeneca has announced a significant $445 million expansion of its Texas manufacturing facility, aimed at doubling the production capacity of its drug Lokelma. This strategic investment is part of the company’s broader initiative to enhance its manufacturing capabilities in the United States, which encompasses a total commitment of $50 billion. The expansion underscores AstraZeneca’s focus on meeting the increasing demand for Lokelma, a treatment for hyperkalemia, as well as its intent to strengthen its overall supply chain resilience.
The context of this expansion reveals a growing trend among pharmaceutical companies to localize production and reduce reliance on overseas manufacturing. As regulatory pressures and supply chain disruptions continue to challenge the industry, AstraZeneca’s move reflects a proactive approach to ensure consistent product availability and compliance with stringent quality standards. This expansion not only positions AstraZeneca to better serve the North American market but also reinforces its commitment to innovation and sustainable growth in the pharmaceutical sector.
The implications of this investment are significant for the B2B pharmaceutical landscape. By increasing its production capacity, AstraZeneca is likely to enhance its competitive edge in the market, potentially influencing pricing strategies and availability of Lokelma. Furthermore, this expansion may prompt other companies to reevaluate their manufacturing strategies, particularly in the context of regulatory compliance and supply chain integrity, as they navigate an increasingly complex global market.
Get started today with Solo access →