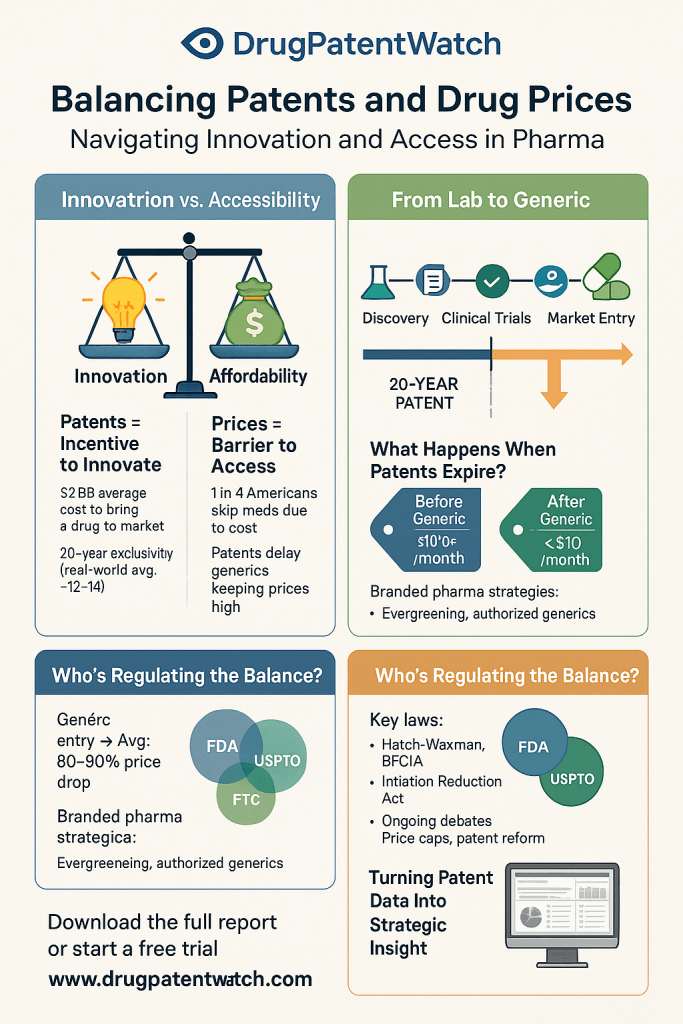
The U.S. Food and Drug Administration (FDA) has granted approval for Ascendis Pharma’s Yuviwel, a weekly treatment for pediatric patients with growth hormone deficiency due to dwarfism. This approval introduces a significant advancement in the management of this condition, providing a more convenient dosing schedule compared to the existing daily injection option, Voxzogo by BioMarin.
This shift to a weekly administration could enhance patient compliance and ease the burden on caregivers, addressing a critical need in the treatment landscape for pediatric dwarfism. With Yuviwel’s approval, healthcare providers now have an alternative that may improve the quality of life for affected children and their families.
The implications of this approval extend beyond patient convenience; it signals a competitive shift in the market for growth hormone therapies. As companies like Ascendis Pharma innovate to meet patient needs, stakeholders in regulatory, quality assurance, and supply chain sectors must adapt to the evolving landscape and consider the potential for increased market dynamics.
Open the full market picture for your next decision →