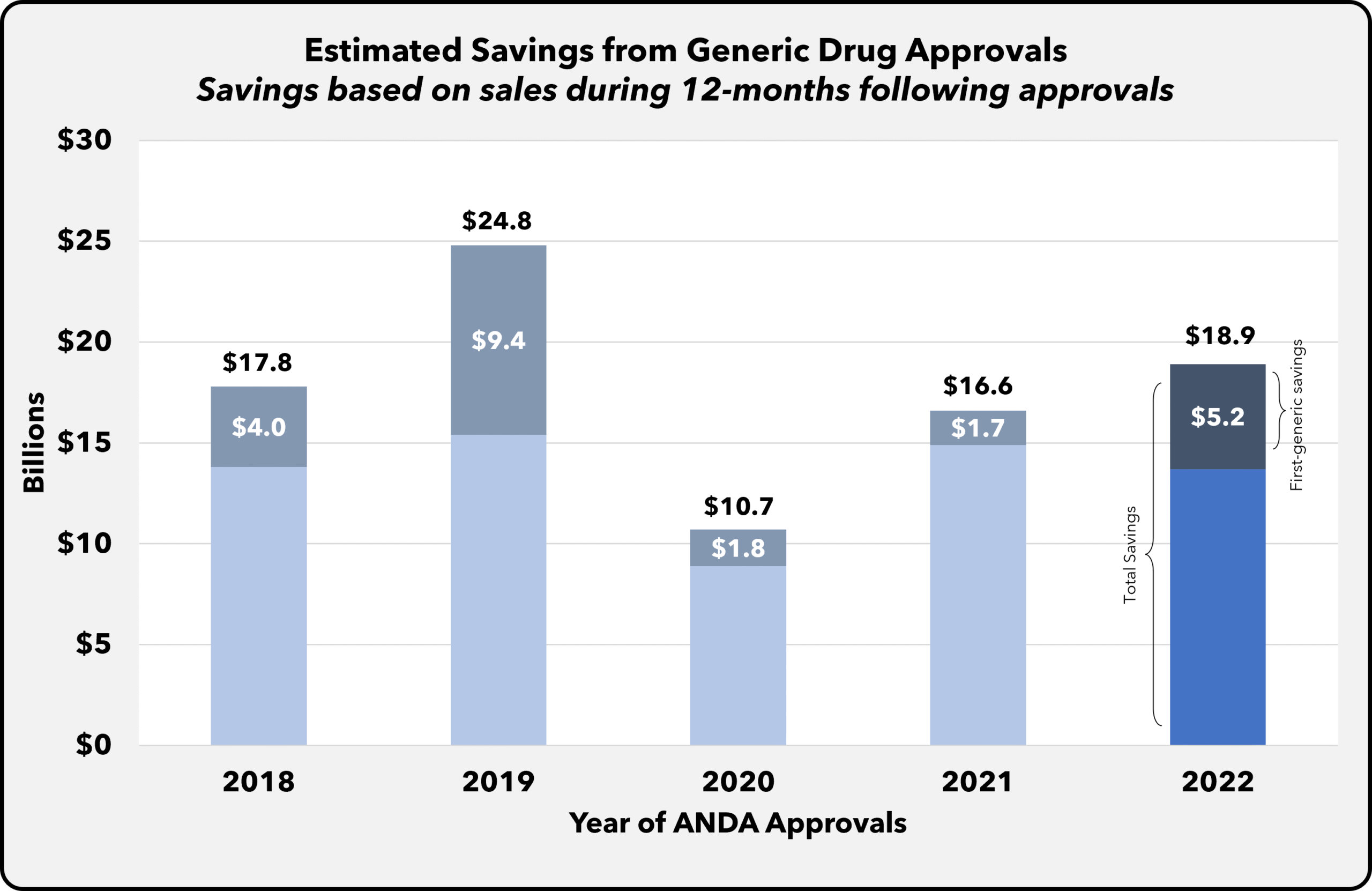
The FDA’s Center for Drug Evaluation and Research (CDER) has recently granted approvals for the first generic versions of several established medications, marking a significant milestone in the pharmaceutical landscape. These approvals not only validate the rigorous scientific and regulatory framework established by the FDA but also underscore the agency’s commitment to fostering competition in the market.
As the pharmaceutical industry continues to grapple with rising drug costs, the introduction of these generic alternatives is poised to provide healthcare providers and patients with more affordable treatment options. By ensuring that high-quality generics are available, the FDA is facilitating broader access to essential medications, which can ultimately lead to improved patient outcomes.
This development has important implications for various stakeholders in the pharma sector, including regulatory professionals, quality assurance teams, and sourcing specialists. It emphasizes the need for ongoing collaboration between manufacturers and regulatory bodies to expedite the approval process for generics, thereby enhancing the overall efficiency of the drug supply chain.
Use the database as your supply chain compass →