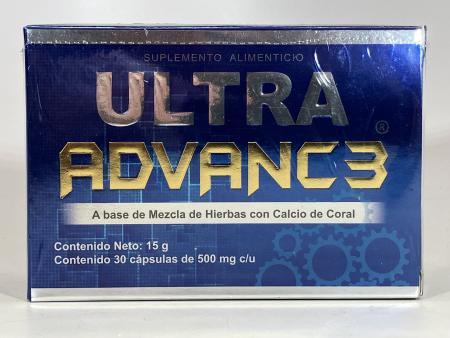
The Food and Drug Administration (FDA) has issued a warning against the use of ULTRA ADVANC3 and ULTRA ADVANC3 GOLD, citing concerns over hidden drug ingredients that may pose health risks. These products, marketed for joint pain relief, are available on various online platforms, including Amazon and Naturista Rex, as well as potentially in retail outlets.
This advisory underscores the ongoing challenge of ensuring product safety in the dietary supplement market, where regulatory oversight can be limited. The presence of undisclosed substances not only jeopardizes consumer health but also raises significant implications for manufacturers regarding compliance and liability. Stakeholders in the pharmaceutical and dietary supplement industries must remain vigilant, as the FDA’s actions highlight the critical need for transparency and rigorous quality assurance practices to protect consumers and maintain trust in the marketplace.
Get started today with Solo access →