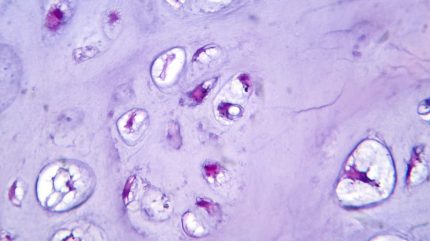
Inhibrx has announced plans to submit its bone cancer drug to the FDA next year, following the release of compelling data from its recent clinical trials. The results, unveiled Thursday, demonstrate significant efficacy across a spectrum of hard-to-treat tumors, marking a potential breakthrough in the oncology landscape.
This development is particularly noteworthy as it highlights the ongoing innovation within the biotech sector, where companies are increasingly focusing on targeted therapies for complex malignancies. The promising data not only strengthens Inhibrx’s position in the competitive oncology market but also raises expectations for regulatory approval, which could pave the way for new treatment options for patients with limited alternatives.
The implications of this submission extend beyond Inhibrx, as successful outcomes could influence investment trends and research priorities within the industry, prompting other firms to accelerate their own oncology programs in response to the evolving regulatory landscape.
Get started today with Solo access →