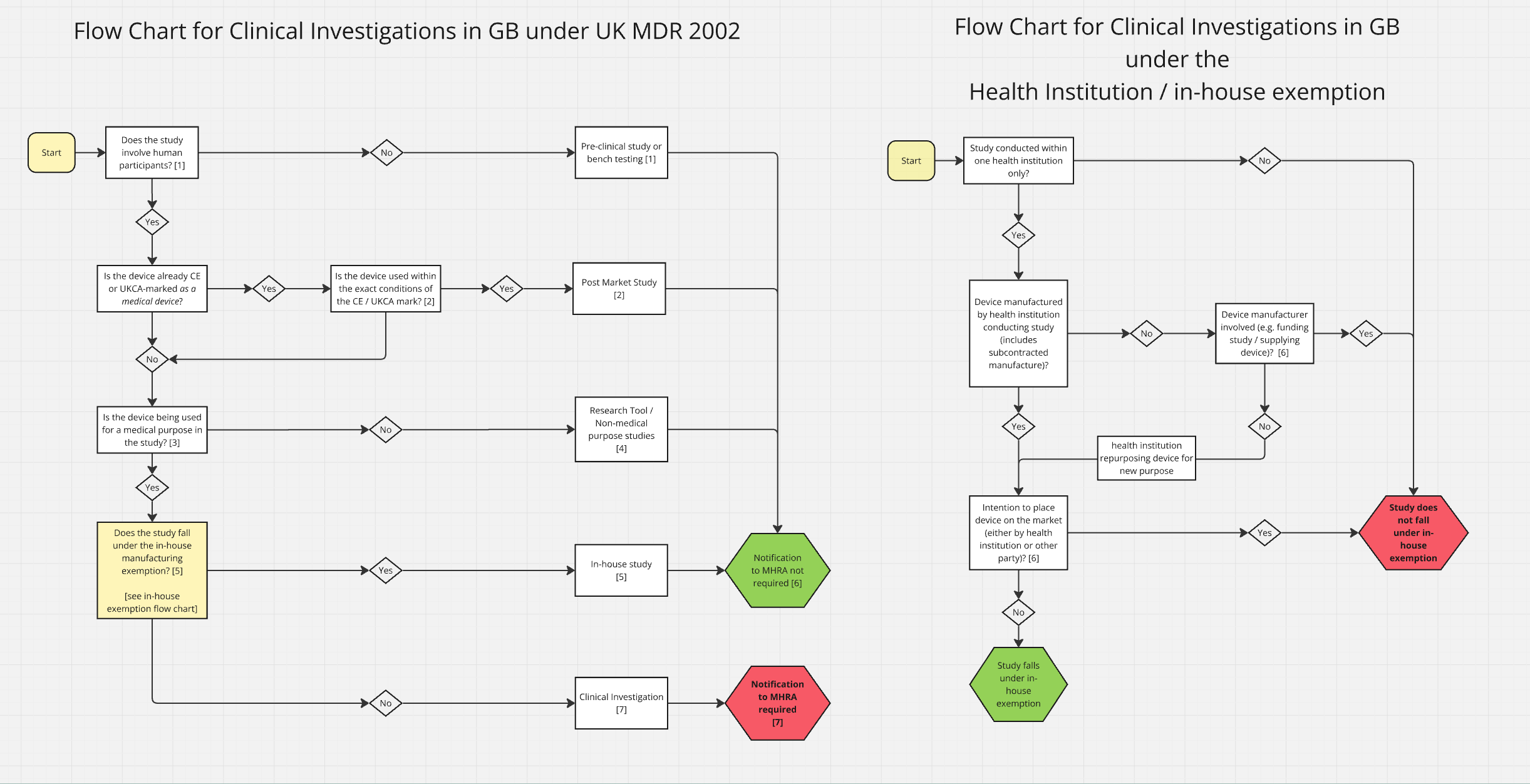
The Medicines and Healthcare products Regulatory Agency (MHRA) has released updated guidance on how to notify the agency of intentions to conduct clinical investigations for medical devices. This development comes in light of increasing scrutiny on clinical trials and the need for enhanced regulatory compliance in the medical device sector.
With the growing emphasis on patient safety and data integrity, the MHRA’s guidance serves as a crucial framework for manufacturers and sponsors. It outlines the necessary steps and documentation required for notification, ensuring that all stakeholders are aligned with regulatory expectations. This is particularly pertinent as the industry navigates the complexities introduced by recent legislative changes.
The implications of this guidance are significant for B2B professionals in the pharmaceutical sector, particularly those involved in regulatory affairs, quality assurance, and clinical operations. Adhering to these guidelines not only facilitates smoother approval processes but also reinforces the commitment to maintaining high standards in clinical research.
Use the database as your supply chain compass →