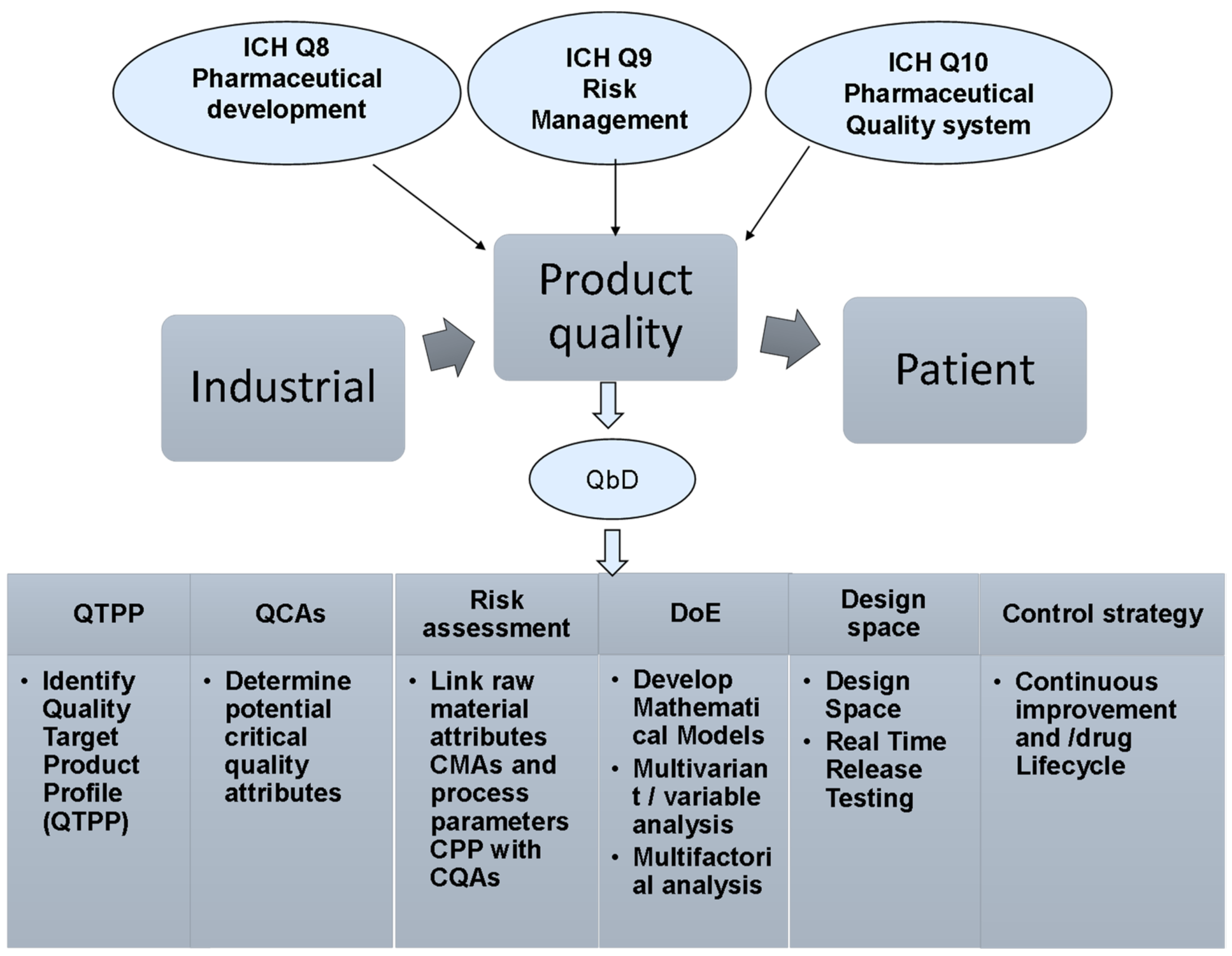
The FDA has granted approval to Organon and Henlius Biotech’s Poherdy, marking it as the first interchangeable biosimilar to Roche’s Perjeta, which is used for treating HER2-positive breast cancer. This significant milestone not only underscores the growing acceptance of biosimilars in oncology but also reflects the ongoing efforts to enhance patient access to effective therapies.
The approval of Poherdy is particularly relevant in the context of rising healthcare costs and the increasing demand for affordable treatment options. By providing a biosimilar that can be used interchangeably with Perjeta, Organon and Henlius are poised to impact the market dynamics significantly, potentially leading to lower prices and increased competition among manufacturers.
The implications of this approval extend beyond market competition; it also highlights the regulatory landscape’s evolving nature regarding biosimilars. As more biosimilars gain approval, stakeholders in the pharmaceutical industry, including regulatory bodies, QA/QC professionals, and sourcing teams, will need to adapt their strategies to navigate this changing environment effectively.
Use the database as your supply chain compass →