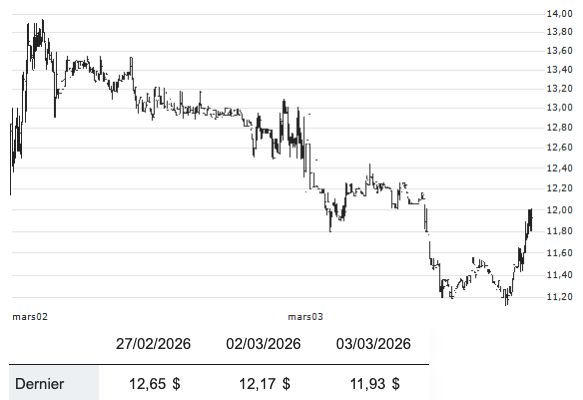
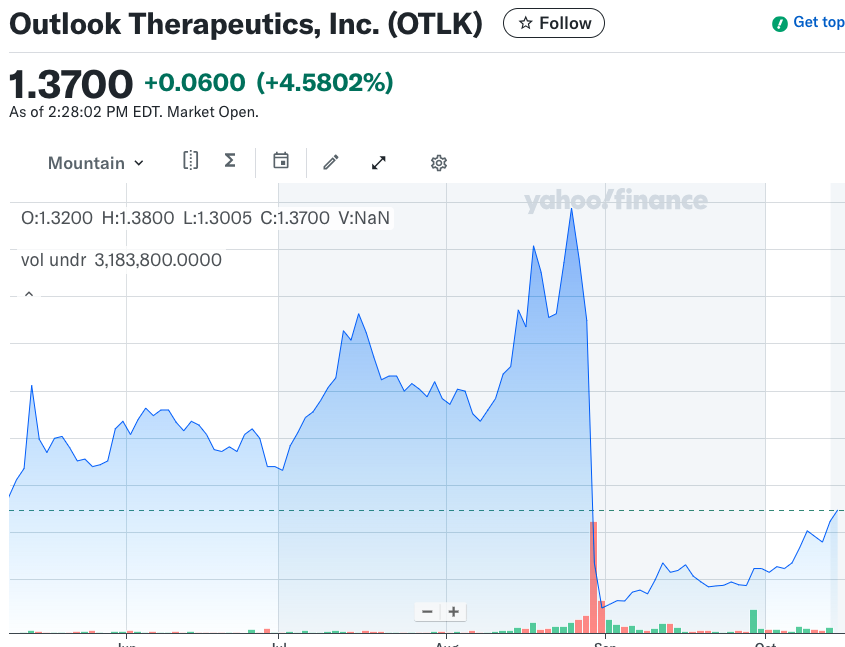
Pharma Rings the Changes as CEO Shake-Up Continues
Recent shifts in leadership across the pharmaceutical sector have highlighted a growing trend of CEO departures, with some executives leaving for personal reasons while others are being ousted due to disappointing company performance. This wave of changes reflects the industry’s increasing pressure to deliver results amid evolving market dynamics and regulatory scrutiny. The implications of…