The biotech markets of early 2026 have become a theater of the extreme, where the ethereal promises of silicon-based discovery collide with the cold, carbon-based reality of clinical outcomes. At the center of this storm sits Generate:Biomedicines, a company that embodies the industry’s most intoxicating ambition: the transition from “bespoke” drug discovery to “programmable” biology.

On February 27, 2026, Generate walked a tightrope over the Nasdaq, pulling off a $400 million IPO—the largest the sector has seen since the 2024 rebound. Yet, within hours of the opening bell, the celebration turned into a wake. For those of us who have spent decades tracking the pulse of the clinic, the “Generate Case” is more than a market correction; it is a profound lesson in the price of innovation during an era of “Day Zero” patent cliffs and AI skepticism.
The “Feverish” Sprint to the Ticker
To understand the drama, one must look at the weeks leading up to the listing. CEO Mike Nally and his team reportedly worked “feverishly” through the 2025 holiday season, racing to catch a window of investor appetite that everyone sensed was about to slam shut. Backed by the powerhouse of Flagship Pioneering and a board featuring Nobel laureate Frances Arnold and Moderna’s Stéphane Bancel, Generate was never going to be a quiet debut.

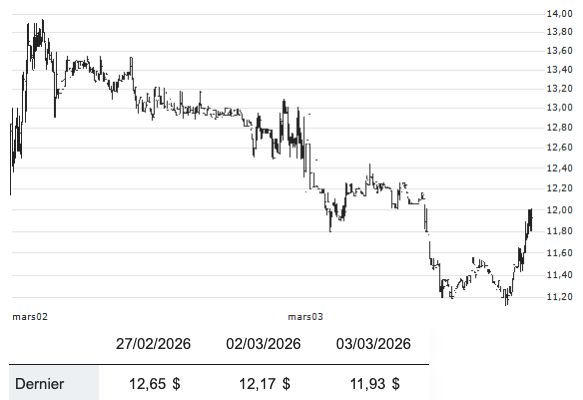
The company priced 25 million shares at $16.00, the midpoint of its range, reflecting a $2.2 billion market capitalization. On paper, it was a triumph of the “AI-native” model—a validation of the Chroma platform and its diffusion-based protein design. But the market’s response was a visceral rejection of the hype. Shares (GENB) opened at $15.00 and plummeted 20.94% by the close of the first day, ending at $12.65.
Why the Bloodshed?
Why did a company with $934 million in total funding and high-profile partnerships with Amgen and Novartis see a fifth of its value erased in eight hours?. The answer lies in the unforgiving arithmetic of the 2026 biotech landscape:
The Burn Rate vs. The Runway: In 2025, Generate spent a staggering $225 million on R&D. Before the IPO, they were staring at a one-year cash runway—a “limited” horizon that signaled to institutional investors that this wasn’t an investment, it was a rescue mission.
The Clinical Anchor: Wall Street is no longer buying “platforms”; it is buying “products.” Generate is the first AI-focused biotech to reach Phase 3, with its lead candidate GB-0895—a long-acting anti-TSLP antibody for severe asthma. While the technology aims for a revolutionary six-month dosing schedule, the SOLAIRIA-1 and -2 trials will not yield full enrollment until 2028. Investors, spooked by a strong Producer Price Index (PPI) report and a broader market sell-off, were unwilling to wait.
The Ghost of Pipeline Volatility: The ghosts of 2025 still haunt Generate. The company was forced to shelve its COVID-19 program (GB-0669) and endure layoffs last year, reminding the market that even AI-designed proteins are not immune to shifting commercial demand.
The Soul of the Matter: Programmable Biology’s Reality Check
For the pharma professional, the Generate drama exposes the “asymmetric risk” of modern drug development. The industry is facing a $200 billion revenue chasm by 2030 as blockbusters like Eliquis lose exclusivity. AI is heralded as the bridge across that chasm, with the potential to double IND success rates.
Yet, the 21% first-day drop is a reminder that “Generative Biology” is still biology. It requires thousands of patients in global multicenter trials like NCT07276724, where 786 participants will determine if Generate’s diffusion models can truly predict human immunology.
Generate remains a “high-risk, high-reward” titan. They are sitting on $300 million earmarked specifically for those Phase 3 trials—a war chest that ensures the experiment continues. But the emotional toll on the sector is undeniable. The day Generate went public was supposed to be the “coronation” of AI in the clinic. Instead, it became a reminder that in the biotech cathedral, the ultimate deity remains the clinical data, and it does not bow to the speed of the algorithm.
As we watch GENB navigate its first quarter as a public entity, the question isn’t just whether GB-0895 will work. It is whether the public markets have the stomach for the multi-year, multi-billion-dollar gestation period of the next generation of medicine. For now, the drama in Somerville continues, and the rest of the industry is watching with bated breath—and tightened purse strings.
Open the full market picture for your next decision →