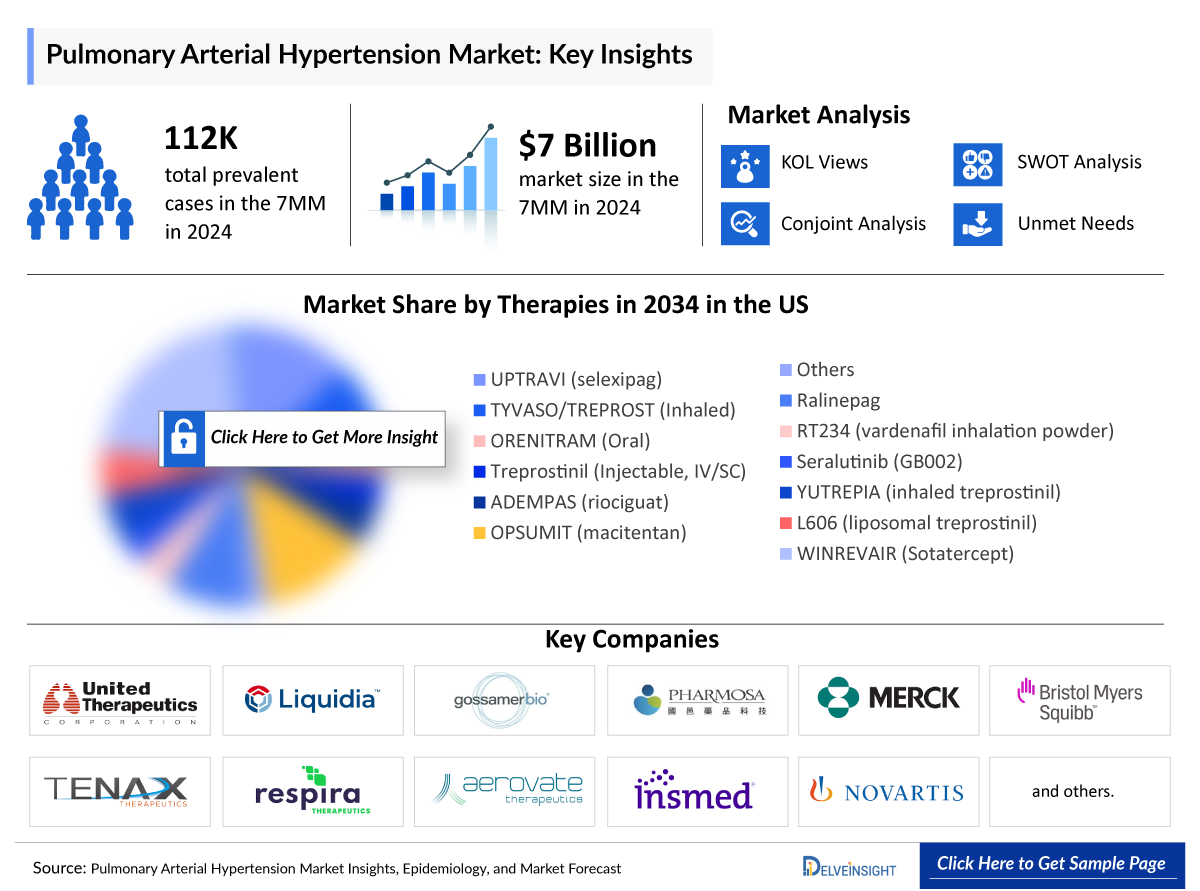
United Therapeutics has announced that its investigational drug ralinepag has successfully completed a Phase 3 clinical trial, positioning the company to submit a New Drug Application (NDA) to the FDA within the year. This milestone not only reflects the drug’s efficacy in treating pulmonary arterial hypertension (PAH) but also underscores the company’s commitment to addressing unmet medical needs in this therapeutic area.
Ralinepag, if approved, is anticipated to be launched in 2027, which could significantly alter the treatment landscape for PAH patients. The success of this drug could enhance United Therapeutics’ portfolio and reinforce its standing in the competitive market for PAH therapies, where effective treatment options are critically needed. The implications of this development extend beyond United Therapeutics, as it may stimulate further innovation and investment in pulmonary hypertension therapies.
Get started today with Solo access →