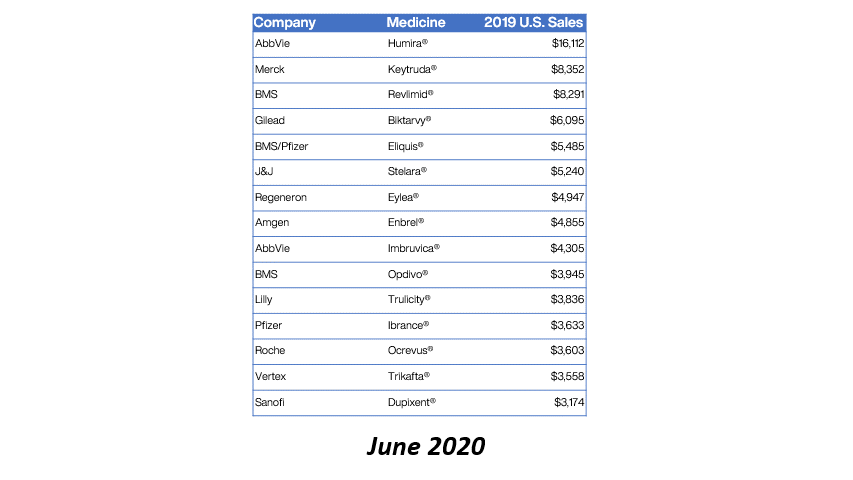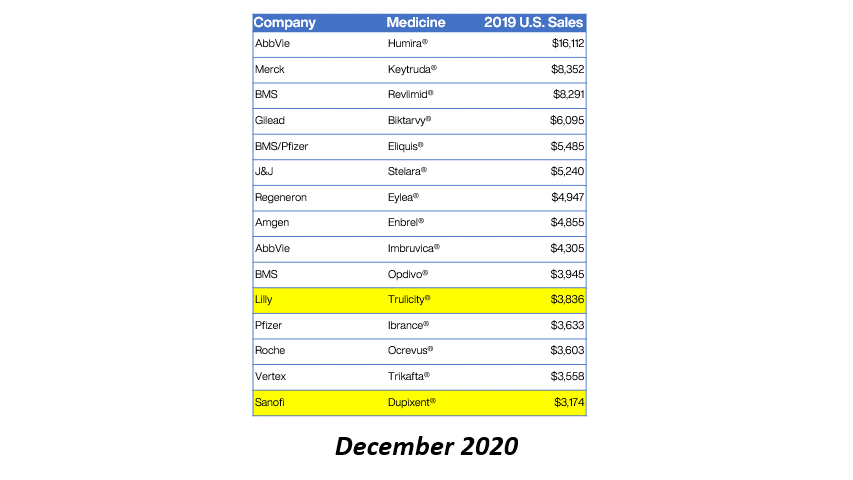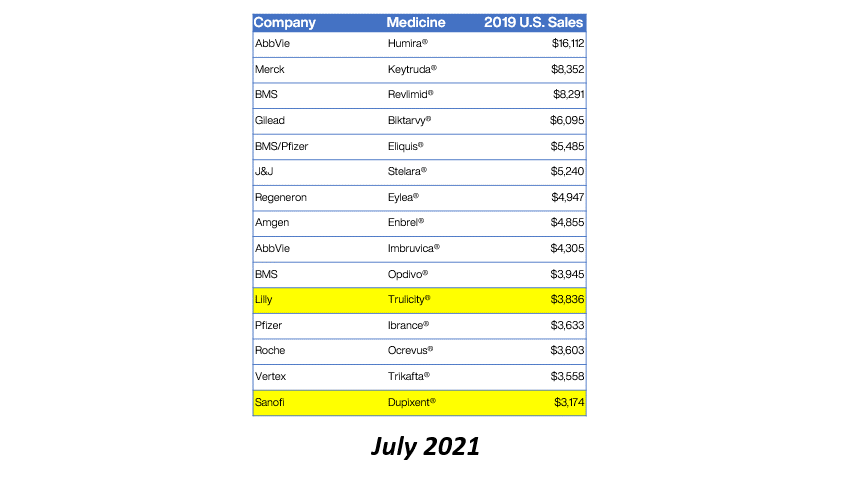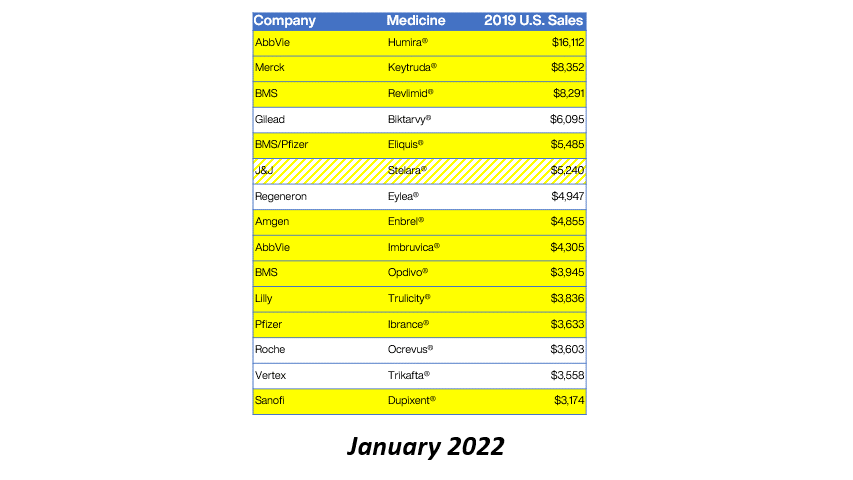
Bristol Myers Squibb (BMS) has received approval from the European Commission for Sotyktu (deucravacitinib) as a treatment for active psoriatic arthritis (PsA). This milestone not only enhances BMS’s portfolio in the immunology space but also signifies a growing recognition of targeted therapies in managing chronic inflammatory conditions. PsA, affecting a significant portion of the population, often leads to debilitating joint pain and skin lesions, necessitating effective treatment options.
The approval of Sotyktu is particularly timely, as the demand for innovative therapies in the rheumatology sector continues to rise. With its unique mechanism of action as a selective TYK2 inhibitor, Sotyktu positions itself as a promising alternative to existing treatments, potentially improving patient outcomes. This development may also influence market dynamics, prompting competitors to accelerate their own product pipelines in response to BMS’s strategic advancements.
Start your 7-day trial and see what the database can do →