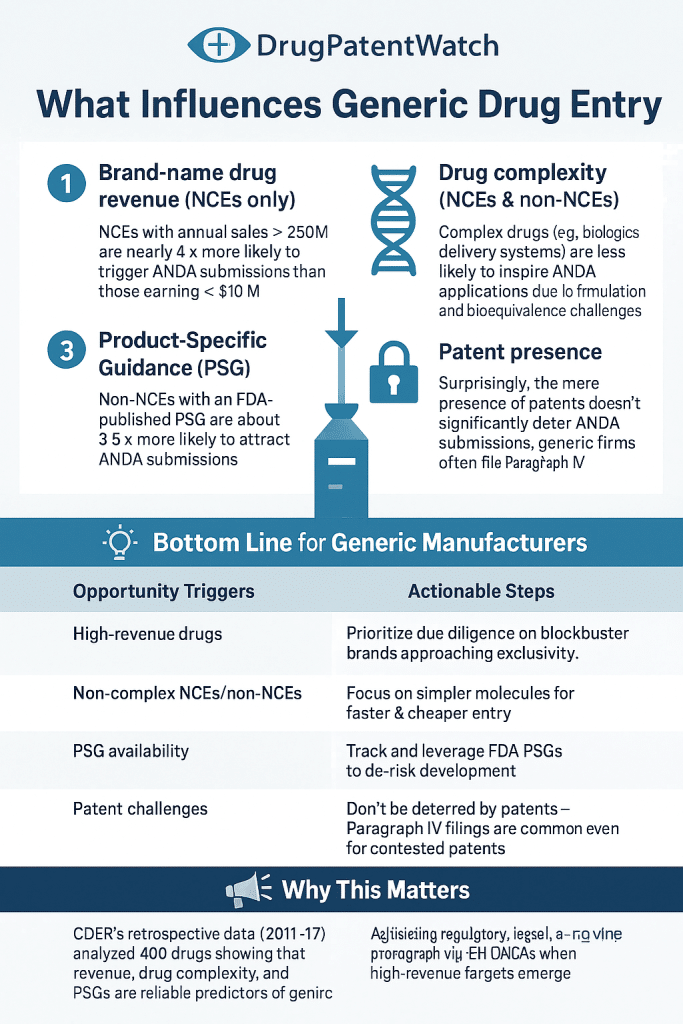
The Generic Drug User Fee Amendments (GDUFA) have been updated to streamline the approval process for generic drugs, significantly impacting the pharmaceutical landscape. These amendments introduce a revised fee structure aimed at increasing the efficiency of the FDA’s review process, thereby expediting the entry of affordable generic medications into the market.
In the context of rising healthcare costs and the ongoing push for accessible treatments, GDUFA’s enhancements reflect a strategic response to industry demands for faster approvals and reduced bottlenecks. By facilitating a more efficient review process, the FDA aims to bolster competition among generic manufacturers, ultimately benefiting patients and healthcare systems alike.
The implications for pharma B2B professionals are substantial, as the revised fee structure and payment methods may influence budgeting and financial planning within regulatory, QA/QC, CMC, and sourcing departments. Stakeholders must stay informed about these changes to effectively navigate the evolving regulatory landscape and capitalize on new opportunities.
Open the full market picture for your next decision →