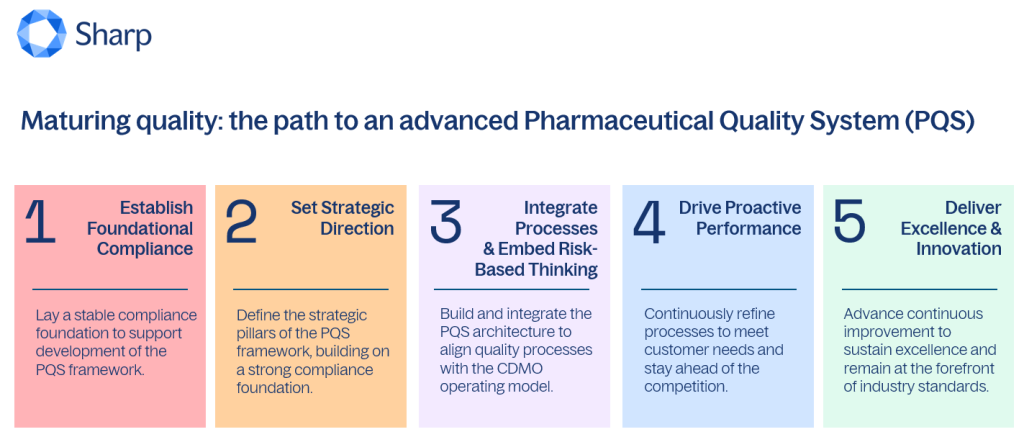
Every quality system begins with the essential aim of achieving foundational compliance, yet the potential for advancement extends far beyond this initial stage. Contract Development and Manufacturing Organizations (CDMOs) are increasingly recognizing the need to evolve their Pharmaceutical Quality Systems (PQS) to meet the demands of a rapidly changing regulatory landscape and heightened market expectations. The journey towards an advanced PQS involves five critical stages, each designed to enhance quality assurance and control processes while fostering a culture of continuous improvement.
This evolution is not merely a regulatory necessity; it is a strategic imperative that can significantly impact a CDMO’s competitiveness and operational efficiency. By adopting a more sophisticated PQS, organizations can better align with industry best practices, reduce risk, and ultimately deliver higher quality products to their clients. As the pharmaceutical sector continues to navigate complexities, the commitment to advancing quality systems will be a defining factor in the success of CDMOs moving forward.
Use the database as your supply chain compass →