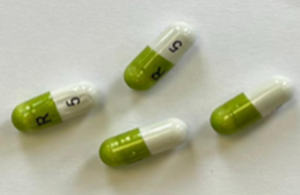
The Medicines and Healthcare products Regulatory Agency (MHRA) has issued a precautionary recall for a specific batch of blood pressure medication following a significant manufacturing error. This directive instructs pharmacy and healthcare professionals to cease the distribution of the affected batch and to return all remaining stock to suppliers. Such recalls are critical in maintaining patient safety and ensuring that healthcare providers are not inadvertently dispensing compromised products.
This incident underscores the ongoing challenges within pharmaceutical manufacturing, where adherence to stringent quality control measures is paramount. The implications of this recall extend beyond immediate patient safety concerns; they also highlight the potential for regulatory scrutiny and the need for enhanced oversight in the manufacturing processes. As the industry grapples with these issues, it is essential for professionals in regulatory, QA/QC, and CMC roles to prioritize compliance and risk management strategies to prevent future occurrences.
Start your 7-day trial and see what the database can do →